Physical and Chemical Properties of Water: A Detailed Scientific Guide
Published on 26-02-2026 By Dr Altaf
Around 70% of the land is covered with water. Life itself totally depends on its existence, it is important to understand thoroughly, and for that, what properties water possesses and how it affects life on Earth. For example, if you are a learner and want to know about the physical and chemical characteristics of this amazing liquid.
In this scenario, this blog will help you understand the fundamental physical and chemical properties of water, molecular bonding and the states in which it exists in different environments. Water not only holds the spiritual values, the physical and chemical aspects help us to understand the complete properties why the existence of this element is essential for life itself.

Chemical Composition and Definition Of Water
It is an important topic to ask before getting into the details. In basic terms, the special qualities that make it such an essential element on Earth depends on the weak molecular bonding between Hydrogen H2 and Oxygen O. The chemical and physical aspects make water a substance that not only inhibits life but also maintains its importance even after its creation.
The Molecular Geometry Of Water
The structure of water (H₂O) clearly shown in the diagram below
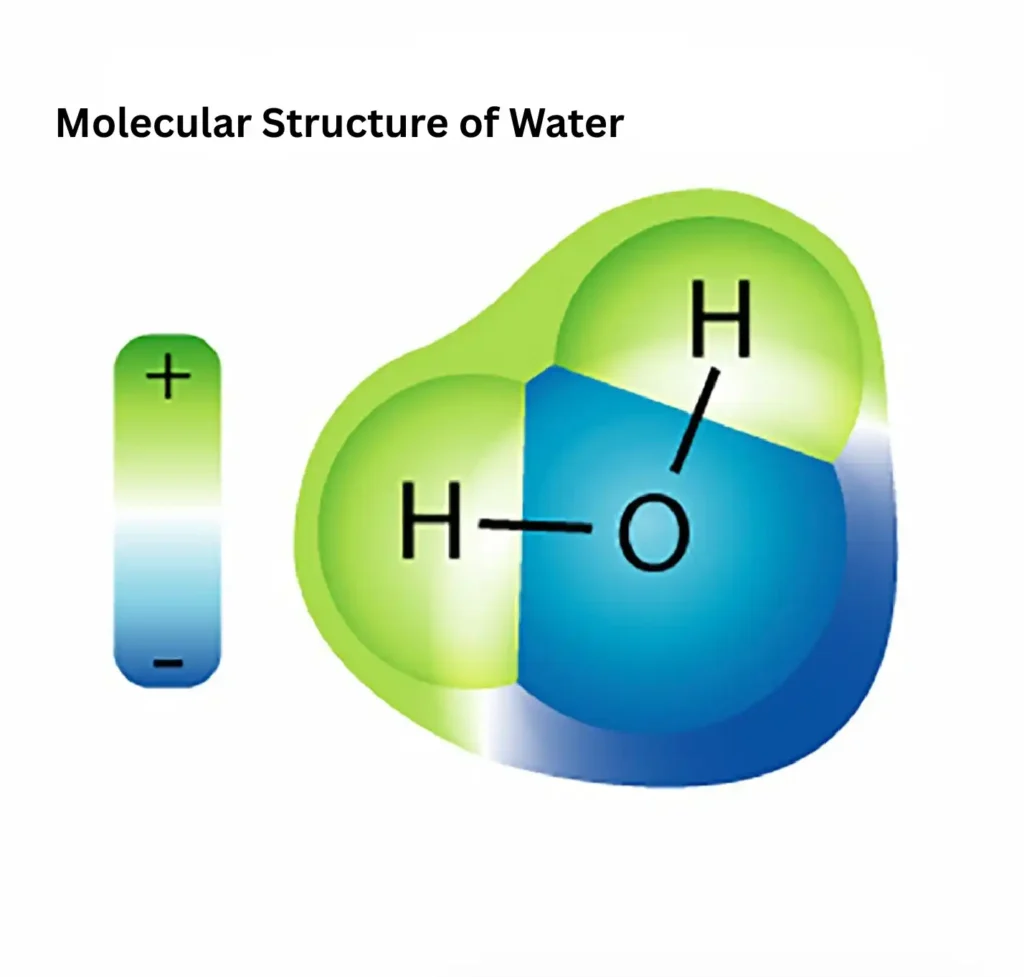
If you’re curious about how water molecule structure relates to it’s classification in chemistry,check out our article on Is water Organic or Inorganic? for your understanding.
Polarity of Water and Hydrogen Bonding
Water as Polar Molecule
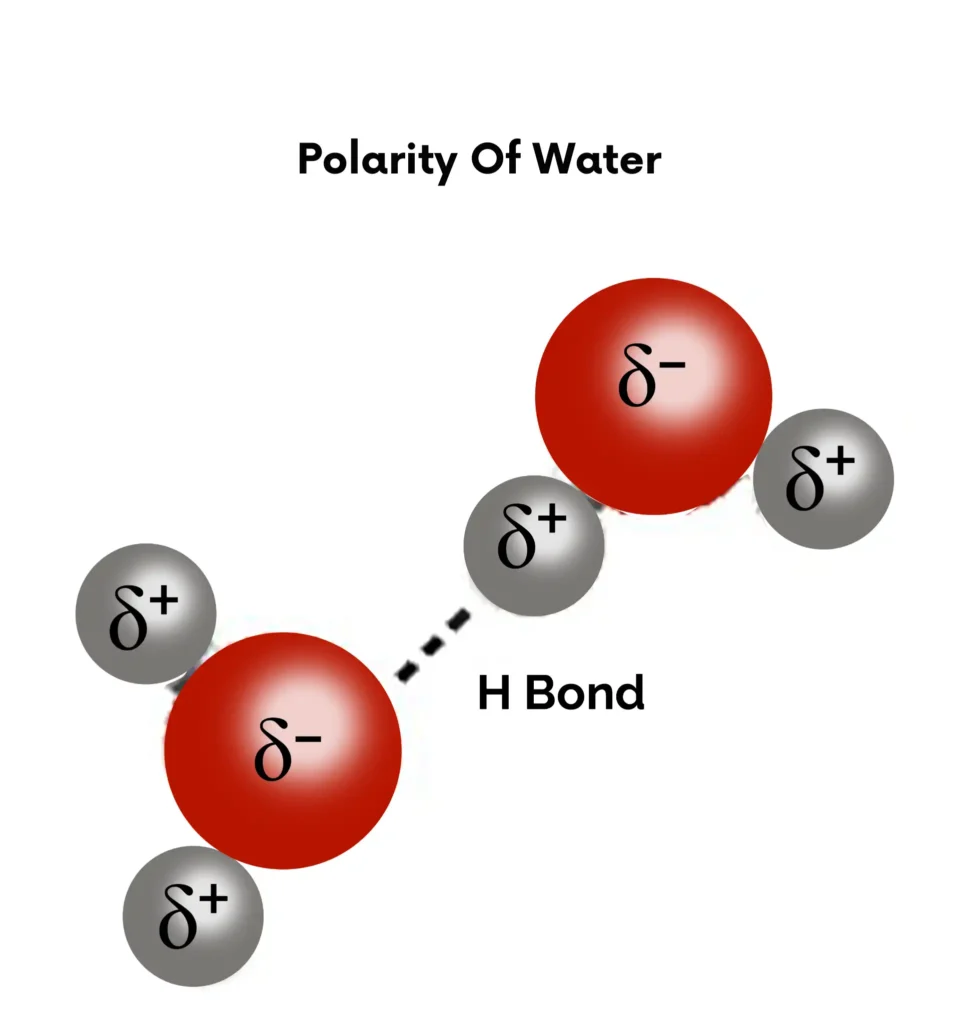
Natural Cohesion and Adhesion Examples:
Cohesion means water drops stick together – just like morning dew clings to blades of grass.
Water climbing through a plant’s stem to reach its leaves happens because of the stickiness between water and the tube walls.
Key Physical Properies of Water
One of the most unique physical property of Water, it looks clear, smells nothing, tastes flat – yet boils and melts at surprisingly high temps. Its skin pulls tight, letting bugs walk on it instead of sinking through. Found frozen, flowing, or floating around us daily because it shifts forms so easily.
Colorless,Odorless and Tasteless
Pure Water has no color,taste and smell when present in small amount.It allows light to pass through without distortion,it is ideal for cooking.Assume no contaminants in water making it odorless.Any taste in water come from dissolved minerals and pollutants.
Density Of Water and Ice
The way water shrinks when freezing stands out as one of its weirdest traits. Unlike nearly everything else, it becomes denser as it cools down. When frozen, ice ends up on top since the molecules line up in a rigid pattern.
Compressibility
The compressibility of water is tiny – it shrinks slightly when squeezed hard. Because it barely gets compressed, pressure moves through it easily, whether in machines or nature.
High Heat Capacity
Water soaks up plenty of heat without getting hotter fast – thanks to its unusually high ability to hold energy. Thanks to traits like this, it plays a key role in keeping life going on Earth. Creatures living underwater benefit from steady warmth due to this trait.
Lakes or oceans soak up heat slowly because they’ve got so much water. That helps keep the planet’s weather steady. Without this, shifts in temperature could get way worse.
High Heat Capacity: Why Is It Important?
- Keeping big heat swings down keeps nature steady.
- Keeping Earth’s weather in check matters a lot.
High Heat of Vaporization
Water also requires a significant amount of energy to turn into vapor – it doesn’t simply transition from liquid to gas easily. Since sweat pulls heat away as it dries off your skin, this feature helps living things manage their warmth by sweating.
People chill down by losing heat when sweat dries off their skin.
Surface Tension
Water sticks together well, so it pulls tightly at the surface. With nothing above, surface molecules grip each other more strongly. This pull lets water bead up, also letting bugs skate across without sinking.
Specific Heat of Water
The specific heat of water means how much energy you need to warm up one gram by just one degree. Because it takes quite a bit – around 4.18 joules per gram – it can soak up warmth without getting hot fast. This ability keeps nature’s temperatures more stable, from oceans to animals. So bodies and climates don’t swing wildly when heated or cooled.
Vapour Pressure of Water
Vapour pressure happens when water turns to gas and pushes against its surroundings at a set temp. When things get warmer, that push gets stronger – so more water slips into the air. Because of this shift, liquid vanishes faster or boils more quickly under heat.
Triple Point of Water
At exactly 0.01°C and 611.657 pascals, water turns into ice, stays liquid, or becomes vapor – all at once. This unique mix of heat and air squeeze marks where all forms meet. Scientists rely on it when studying energy flow. Pure water hits this spot without any added stuff.
Chemical Properties and Behavior of Water
Water might mix up in chemical actions, taking part in different changes – like in living things, nature scenes, or factory setups.
Self-Ionization
Water can turn into ions when two molecules bump into each other, making H₃O+ and OH⁻. This happens on its own in clean water – key to how we measure acidity.
Acidity Nature of Water
The acidity of water refers to how well it can dissolve gases – such as carbon dioxide – that create a mild acid. Because of this trait, living things in water can survive.
Acid-Base Reactions
Water can behave like an acid or a kind of base when reacting – it depends on the situation. Sometimes it gives away a hydrogen ion; other times, it picks one up instead. Because of this switch, it helps cancel out strong acids or alkalis. That’s how it keeps things chemically steady.
Redox Reactions
Water behaves as either an oxidizing agent or a reducing agent. Take rust formation or plant light conversion – it plays a role there.
pH and Water
pH tells you the amount of H⁺ ions in water. Though pure water stays right at 7, adding acidic or alkaline stuff moves that value – hinting if it’s more lemony or slippery.
Chemical Reactions Involving Water
Water takes part in plenty of chemical changes – like when it adds to molecules, breaks them apart, or helps form new ones. Sometimes it’s what gets used up, sometimes it shows up later, other times it just hangs around making things easier.
Hydrolysis Reaction
Hydrolysis is a reaction where water breaks down a compound, typically splitting it into two or more smaller substances. It is common in digestion.
Water as a Solvent
Water can split certain chemicals apart during hydrolysis, forming simpler parts. This kind of change often happens when your body digests food.
Unique Properties of Water in Everyday Life
Beneath this line, you’ll find how water acts differently in daily situations – its odd traits pop up around us without notice.
Why Ice Floats on Water
Ice stays on top since it’s lighter than water. As things cool down, the molecules line up in a pattern that spreads them out. That little gap between them makes ice less dense. Even being frozen, it fills more space – so it ends up feeling easier to lift. It just sits above the liquid instead of sinking.
Why Water Has High Surface Tension
Water sticks together tightly because of hydrogen bonds between its parts. This makes the top act like a thin skin, letting light things sit on it without sinking. Also, this trait helps water bunch up into tiny beads instead of spreading out flat.
Role Of Water in Weather and Climate
Water might shift how weather acts because of its role in climate patterns by means of evaporation, mixing with air conditions instead of staying separate, evaporation, then condensation, followed by precipitation. It holds heat, later releases it – this keeps Earth’s climate steady while helping natural systems thrive.
Water in Cooking and Cleaning
In cooking, water boils things, it steams them, and it also helps dissolve bits inside. When cleaning, it pulls grime apart – its natural pull on gunk lifts messes out, rinsing them off instead.
Unique Features of Water final words
Thanks to unique traits, water’s an incredible substance vital for living things. Its features – say, how dense it is or how it beads up – mix with chemical behaviors like sticking to molecules or dissolving stuff, letting life thrive in many forms.
The way water works in nature makes one thing obvious – life wouldn’t exist like this if water didn’t have its special traits.
Frequently Ask Question
- It helps dissolve stuff so chemical processes can happen.
- It keeps living things plus their surroundings at the right heat level.
- It helps move nutrients around while carrying out waste in living things.


